
The K-values taken from the DePriester Chart for the. Phase Equilibrium (Alternate Form VLE) Historically, when estimates were done by hand: Sometimes the K values are nearly composition independent “hand” techniques of design/solution have used DePriester Charts (hydrocarbons):ĭePriester Chart P = 2 bar T = 100 oC Isobutane others…. ample, near a critical point in the phase diagram where the. Ideal Behavior 2.5. A chart was provided for each hydrocarbon, in which K-value could be obtained by knowingT andPof thesystem. Phase Equilibrium (Alternate Form VLE) Historically, when estimates were done by hand: Historically, K-values for light hydrocarbons were often given in a set of charts constructed by DePrister, then called Depriester charts. ↑ "Define and use K values, relative volatility, and x-y diagrams".Unit Operations Handbook: Volume 1 (In Two Volumes). ↑ "Pbub and Pdew calculation based on the DePriester chart for a fast pyrolysis and hydrotreating process".Read this K-value off the chart (approximately 21.3). Note where the line crosses the methane axis.Ideal Behavior 2.5.5 Equilibrium relation: K-value. Connect the points with a straight line. charts constructed by DePrister, then called Depriester charts.These nomograms have two vertical coordinates, one. DePriester in an article in Chemical Engineering Progress in 1953.

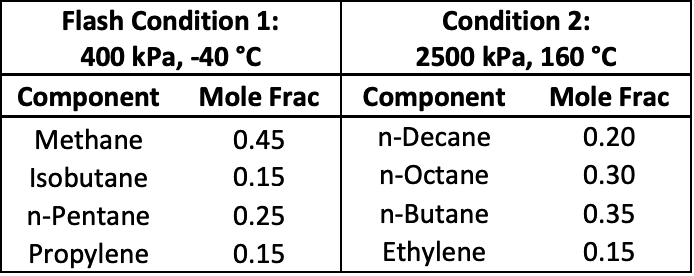
I have attached two diagrams for your reference. It may be approximate, but I really recommend that you try it by hand. DePriester Charts provide an efficient method to find the vapor-liquid equilibrium ratios for different substances at different conditions of pressure and temperature. You may like to try it by hand using the classic DePriester Charts.
